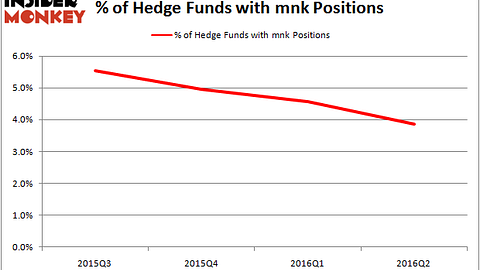
Ironwood Pharmaceuticals, Inc. (NASDAQ:IRWD) has a PDUFA date of February 9, 2017 for the the company’s 72 mcg dose of its chronic idiopathic constipation (CIC) candidate, Linzess (linaclotide). The drug is already approved in its present indication, but currently, because of its high dose (145 mcg), it is not being used to treat more mild cases of the condition.
Linaclotide 72 mcg dose’s original sNDA acceptance was announced June 9, 2016. Why is this all so significant? Well, the company’s biggest news of last year was related to this drug, Linzess. U.S. net sales for the drug are expected to be approximately $625 million for the full year of 2016 (official number not yet released), which represents an estimated growth of more than 35% over 2015, with an estimated commercial margin of greater than 55%.
As for the conditions that linaclotide treats: Irritable bowel syndrome with constipation (IBS-C) and chronic idiopathic constipation (CIC) are gastrointestinal disorders. It’s estimated that as many as 13 million adults in the U.S. may suffer from IBS-C, and as many as 35 million adults in the U.S. may suffer from CIC. IBS-C is generally characterized by hallmark symptoms of abdominal pain and infrequent bowel movements, at less than three times per week.

Copyright: nexusplexus / 123RF Stock Photo
Although CIC is, too, generally characterized by infrequent bowel movements at less than three times per week, there is more to this condition than IBS-C: its symptoms vary – hence the word “idiopathic,” which is when “a disease or condition which arises spontaneously or for which the cause is unknown.” The problem, then, is that the currently available Linzess dose is too potent for as much at 60 percent of sufferers, whose symptoms are either not severe enough or are idiopathic, and the adverse effects don’t outweigh the potential benefits.
Looking closer at the science behind the drug, linaclotide is a guanylate cyclase‐C agonist. Through a host of trials and tests with other GC-C antagonists that the company had designed and developed, linaclotide (out of many contenders) was the drug that Ironwood Pharmaceuticals, Inc. (NASDAQ:IRWD) found worked best. Not only does it reduce gastrointestinal pain, but it also increases fluid secretion in the bowels. It is both of these functions working together that is the key to the massive relief of the primary symptoms of CIC that linaclotide provides.
According to a statement from the company, its primary goals for 2017 are to, one, continue strong Linzess growth and margin expansion and, two, introduce the aforementioned 72 mcg dose. So, not only will the new dose expand the market for Linzess, this 72 mcg “companion” could notably improve Ironwood’s top line going forward: the company has an expected >25% CAGR target on income between 2016 and 2020.
Up to 35 million adults suffer from CIC, meaning IWRD’s 72 mcg Linzess dose could be a real catalyst in helping the company achieve its 2017 goal of increasing commercial market share. If you’d prefer to judge the potential impact from a more straight-forward position, experts say the 72 mcg dose could double the market potential for the drug.
Nothing is certain, of course. While the safety profile of the drug seems sound, the FDA has shocked markets before in its interpretation of low-dose regimen drugs, and clinical benefit is going to have to prove beyond reason if the agency is going to give this one a green light. With that said, however, we don’t think that should be any issue. The higher dose of the drug is generally going to be the one that causes any unforeseen tolerability issues, and that is already regarded as safe from an agency perspective. As such, that the FDA would have any hang ups based on potential safety doesn’t seem realistic.
There is the potential for competition coming in to play, and impacting the company’s topline estimates. We highlighted Synergy Pharmaceuticals Inc (NASDAQ:SGYP) late last week, and its drug Plecanatide. This one has a PDUFA of January 29, and may encroach on Ironwood Pharmaceuticals, Inc. (NASDAQ:IRWD)’s revenue generation potential if it picks up approval.
With that said, we still think there is considerable upside for the company on a nod for commercialization, and this makes it very much one to watch heading into decision day. Definitely one to watch near term, and one to keep an eye on throughout the remainder of 2017 to see if the company can live up to its own expectations.
Follow Ironwood Pharmaceuticals Inc (NASDAQ:IRWD)
Follow Ironwood Pharmaceuticals Inc (NASDAQ:IRWD)
Receive real-time insider trading and news alerts
Note: This article is written by Mark Collins and originally published at Market Exclusive.